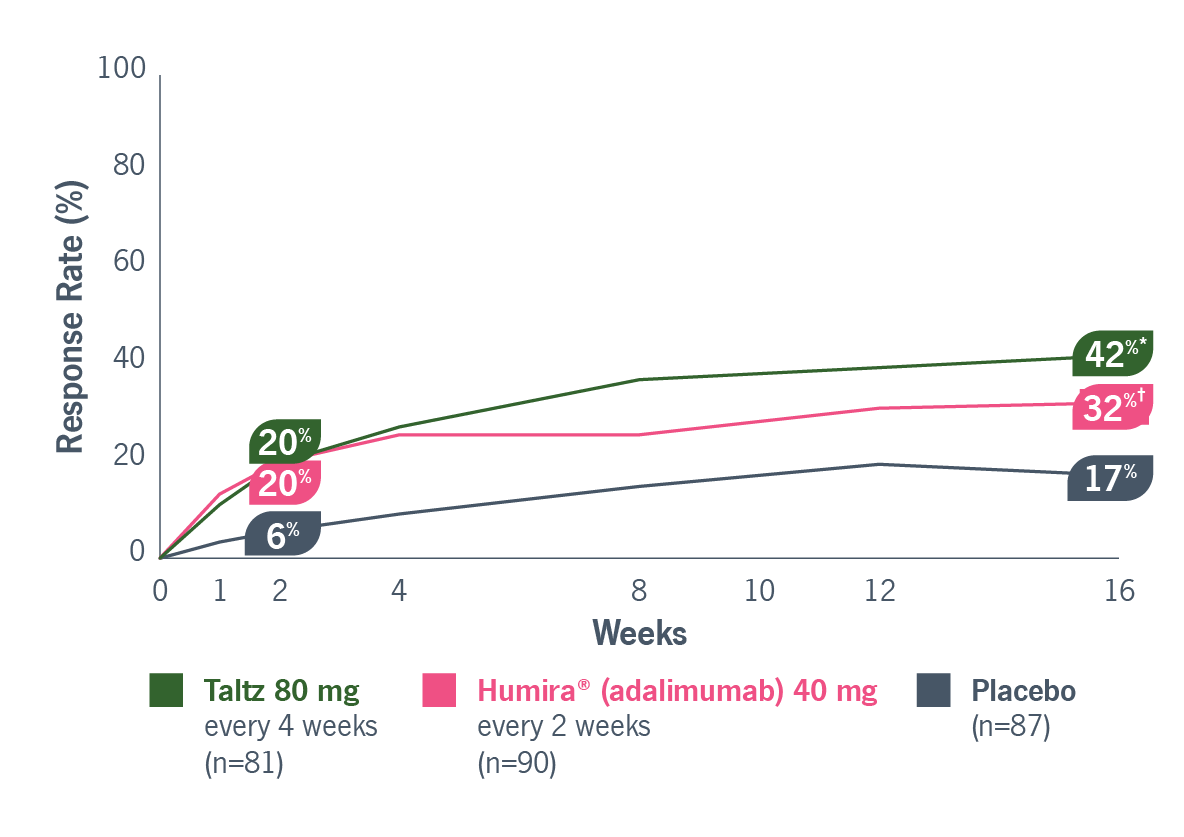
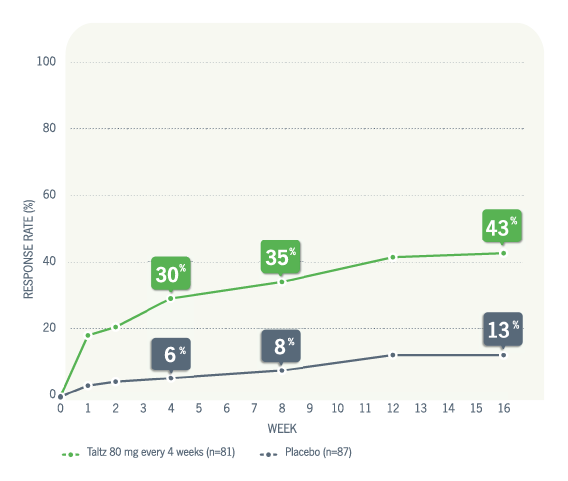
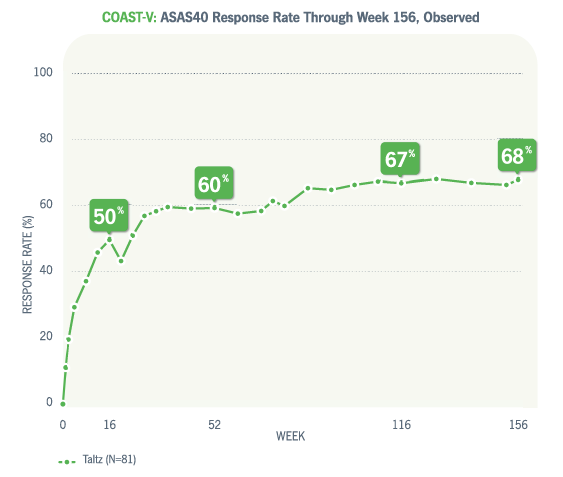
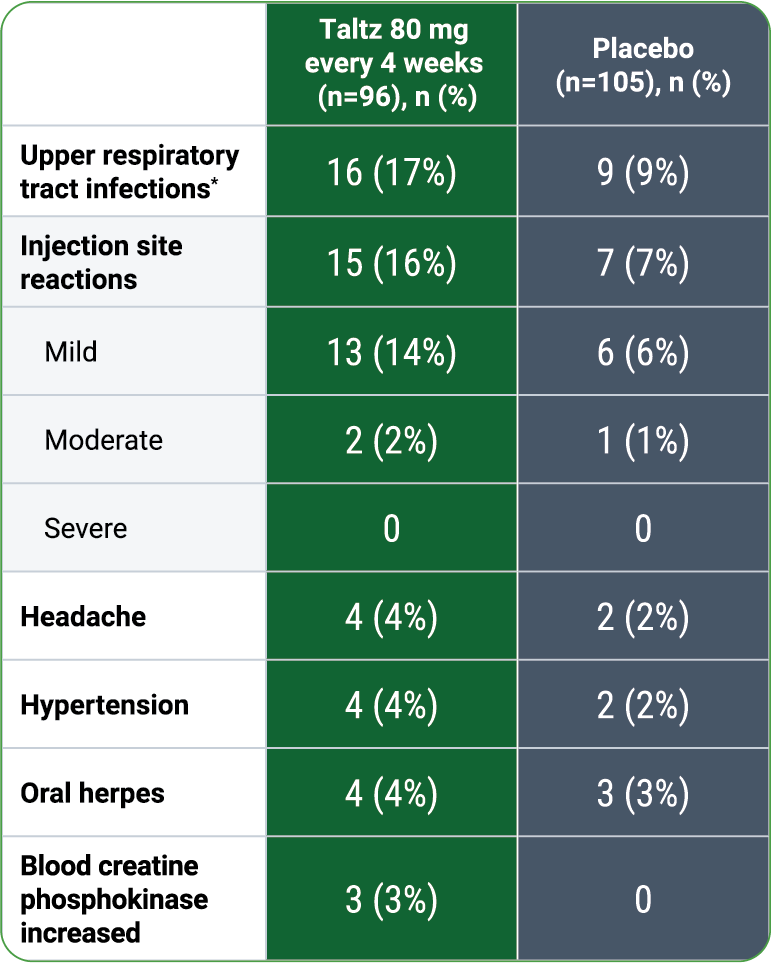
00:00-00:18
[First section of caption fades away and is replaced by second section of caption.]
Caption:
How to identify patients with symptoms of inflammatory bowel disease (IBD) and when to make a referral—Insights from a dermatologist, rheumatologist, and gastroenterologist.
PP-LI-US-0795 01/2024 © Lilly USA, LLC 2024. All rights reserved.
Lilly invited speakers to discuss a specific topic based on a series of questions and assisted in developing a script for discussion. Speakers were encouraged to include their own experience where appropriate. Some of the content in this video may therefore reflect the speakers’ personal opinions and may be specific to their own practice. Particular recommendations are those of the individual physicians and not those of Eli Lilly and Company.
00:19-00:32
[The pictures, names, and captions of the three doctors appear on screen.]
Caption:
Prof. Alexander Moschen, Gastroenterologist, Assistant Professor of Internal Medicine, Consultant Gastroenterologist, Medical University Innsbruck, Austria.
Prof. Dennis McGonagle, Rheumatologist, Professor of Investigative Rheumatology, Consultant Rheumatologist, Leeds Teaching Hospitals NHS Trust, UK.
Dr Joseph Merola, Dermatologist, Associate Physician, Brigham and Women’s Hospital, Associate Professor of Dermatology, Harvard Medical School, Boston, MA, USA.
Dr. Moschen: Hi, my name is Dr. Moschen. I'm joined by Professor McGonagle and Dr. Merola to discuss inflammatory bowel disease from our three different perspectives: gastroenterology, rheumatology, and dermatology.
00:33-00:55
[The caption is replaced by Dr. Moschen’s photograph. A diagram of the intestines with the colon highlighted with the label Ulcerative colitis appears. Then a diagram of the whole intestinal tract appears with the large intestines and then the ileum being highlighted with the label Crohn’s disease.]
Caption:
What is IBD?
Prof. Alexander Moschen, Gastroenterologist
IBD – Ulcerative colitis, Crohn’s disease
Dr. Moschen: IBD is a chronic relapsing-remitting, immune-mediated disorder characterized by inflammation in the gastrointestinal tract. Two major forms of IBD include ulcerative colitis, affecting only the colon, and Crohn's disease, which may affect the whole of the intestinal tract, but typically in a discontinuous region.
00:56-01:15
[A DNA icon within a circle appears, joined by circles representing ulcerative colitis and Crohn’s disease. Then a circle with the words immune-mediated diseases e.g. PsO, axSpA, and PsA appears. come onto the screen as he begins talking.]
Caption:
Ulcerative colitis, Crohn’s disease, immune-mediated disease e.g. PsO, axSpA, PsA
Dr. Moschen: Research shows that there is a lot of overlap in terms of genetic susceptibility between Crohn's disease, ulcerative colitis, and other immune-mediated diseases. We need to keep this relationship in mind and ask our patients with immune-mediated diseases about IBD and vice versa.
01:16-01:35
[A series of people icons appears labeled with the captions, joined by a world map with the United States, Canada, and Europe highlighted.]
Caption:
300 in 100,000; 500 in 100,000, Immune-mediated disorders
Dr. Moschen: If we look at the epidemiology of IBD, a comparable prevalence rate is observed in North America and Europe with approximately 300 to 500 cases per 100,000 inhabitants. By comparison, the incidence rate in patients with immune-mediated disorders is marginally higher.
01:36-02:11
[The caption is replaced by Prof. McGonagle’s photograph. Representative icons labeled with the captions appear and slide off screen to the left.]
Caption:
What clinical symptoms do you look for in patients who may be at higher risk of IBD?
Prof. Dennis McGonagle, Rheumatologist
Abdominal symptoms, Family history, Inflammatory markers, Anemia, Fecal calprotectin
Prof. McGonagle: In rheumatology, we will ask the patients who we suspect might have spondyloarthritis about abdominal symptoms and ask about a family history of IBD. Beyond that, we run routine blood investigations, including inflammatory markers, looking for evidence of anemia, and we may also check fecal calprotectin. And based on these findings, we may then refer the patient to our gastroenterologist to further evaluate for IBD.
02:12-02:44
[A photograph of Dr. Merola appears. Representative icons labeled with the captions appear and slide off screen to the left.]
Caption:
Dr Joseph Merola, Dermatologist
Abdominal symptoms, Family history, Blood, Mucus, Fever, Fatigue, >6-10 bowel movements, hypoalbuminemia, anemia, increased WBC, Fecal calprotectin
Dr. Merola: From a dermatologist perspective, I certainly consider it crucial to obtain the patient’s personal history, in terms of symptoms that might be relevant to IBD as well as family history. I look for red flags such as bleeding, mucus in the stool, fever, and I typically will ask about fatigue, maybe number of bowel movements throughout the day. I would also look for signs in their blood work, such as hypoalbuminemia, anemia, elevated white blood count, as well as considering a fecal calprotectin.
02:45-03:05
[The caption is replaced by Dr. Moschen’s photograph. Then later captions and representative icons animate and slide to the left.]
Caption:
What would be the main questions to ask your patient?
Prof. Alexander Moschen, Gastroenterologist
Have you experienced gastrointestinal symptoms?
Diarrhea, Blood, Mucus, Abdominal pain, >4 weeks
Dr. Moschen: I would recommend asking two simple questions. The first one is, “Have you ever experienced any gastrointestinal symptoms, namely diarrhea, blood in stool, mucus, abdominal pain that have lasted longer than four weeks?”
03:06-03:41
[Captions and representative icons animate and slide to the left.]
Caption:
Does anyone in the family suffer from IBD?
Family history, Abdominal symptoms, Fecal calprotectin
-No further testing (likely IBS)
+Further investigation required (refer to a gastroenterologist)
Dr. Moschen: And secondly, does anyone in the family suffer from IBD? If the patient has a positive family history or is reporting persistent GI symptoms, my recommendation would be to run a test for fecal calprotectin. Fecal calprotectin is the most important parameter to distinguish between inflammatory and functional gastrointestinal diseases and the results are similar to a traffic light system. If the test is negative, it might not really be necessary to do any further testing. Then it's likely IBS.
03:42-04:24
[The caption is replaced by Dr. Moschen’s photograph. Captions and representative icons animate and slide to the left.]
Caption:
How do you differentiate idiopathic gastrointestinal symptoms from real IBD?
Prof. Alexander Moschen, Gastroenterologist
How long have you had these symptoms?
Chronic diarrhea is defined as having symptoms for more than 4 weeks
In patients with immune-mediated disease, refer to a gastroenterologist if GI symptoms persist longer than 2 weeks
How rapid was the onset of symptoms?
Dr. Moschen: The first question to ask is how long have you had these symptoms? The duration of symptoms is very important. IBD is a form of chronic diarrhea and is defined as having symptoms for more than four weeks. In a patient with an immune-mediated disease, I would recommend referring to a gastroenterologist if the patient has GI symptoms for longer than two weeks. Secondly, the onset of symptoms occur very rapidly. It's likely that the problem is not IBD, then it's more likely an acute gastroenteritis caused by virus or bacteria or by food poisoning.
04:25-04:55
[A photograph of Prof. McGonagle appears. Representative icons and captions animate and slide to the left.]
Caption:
Prof. Dennis McGonagle
Rheumatologist
Abdominal symptoms
Bloody diarrhea, Mucus, Weight loss, Colitis, IBS
Prof. McGonagle: From a rheumatology perspective, if a patient’s been on an IL-17 inhibitor for psoriatic arthritis or ankylosing spondylitis for several months and they develop abdominal symptoms, but the absence of bloody diarrhea, mucus, and weight loss and clearly overt colitis and they simply have pains, which may be linked to irritable bowel, we wouldn't jump in and stop treatment.
04:56-05:06
[A photograph of Dr. Merola appears. Representative icons and captions animate and slide to the left.]
Caption:
Dr Joseph Merola
Dermatologist
Abdominal symptoms
Dr. Merola: And I agree, I'd say from the dermatology perspective, abdominal complaints, diarrhea, and GI symptoms are pretty common. So, I wouldn't be in a rush to switch treatment, particularly effective treatment, unless I really saw those true red flags.
05:07-05:27
[A photograph of Dr. Moschen appears. Captions and representative icons animate and slide to the left.]
Caption:
Prof. Alexander Moschen
Gastroenterologist
Idiopathic GI symptoms
IBD
Thank you
Dr. Moschen: So, I fully agree. So, simple questions are really useful tools to begin differentiating idiopathic gastrointestinal symptoms from real IBD. I think it's important to understand the signs and symptoms and how to manage your patients and when to refer to a gastroenterologist. Thank you very much.
05:28-05:35
Caption:
Taltz Label Language
Inflammatory Bowel Disease
Patients treated with Taltz may be at increased risk of inflammatory bowel disease. In clinical trials, Crohn's disease and ulcerative colitis, including exacerbations, occurred at a greater frequency in the Taltz group than the placebo control group. During Taltz treatment, monitor for onset or exacerbation of inflammatory bowel disease and if IBD occurs, discontinue Taltz and initiate appropriate medical management.
05:36-05:40
Caption:
References:
- David T, Ling SF, Barton A. Clin Exp Immunol. 2018;193(1):3-12.
- Egeberg A, Mallbris L, Warren RB, et al. Br J Dermatol. 2016;175(3):487-492.
- Graves NS. Prim Care. 2013;40(3):727-741.
- Juckett G, Trivedi R. Am Fam Physician. 2011;84(10):1119-1126.
- Li WQ, Han JL, Chan AT, Qureshi AA. Ann Rheum Dis. 2013;72(7):1200-1205.
- Ng SC, Shi HY, Hamidi N, Underwood FE, et al. Lancet. 2018;390(10114):2769-2778.
- Silverberg MS, Satsangi J, Ahmad T, et al. Can J Gastroenterol. 2005;19(Suppl A):5A-36A.
-
Taltz US PI. Indianapolis, IN, USA: Eli Lilly USA LLC, 2020.
https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/125521s020lbl.pdf
(Accessed October 14, 2020).
05:41-05:45
Caption:
Abbreviations:
axSpA=Axial Spondyloarthritis
GI=Gastrointestinal
IBD=Inflammatory Bowel Disease
IBS=Irritable Bowel Syndrome
IL-17=Interleukin-17
PsA=Psoriatic Arthritis
PsO=Psoriasis
WBC=White Blood Count
05:46-05:51
Caption:
Eli Lilly and Company would like to thank Prof McGonagle, Dr Merola and Dr Moschen for kindly providing their time and expertise for this video project. Speakers were encouraged to include their own experience where appropriate. Some of the content in this video may reflect the speakers’ personal opinions and may be specific to their own practice.